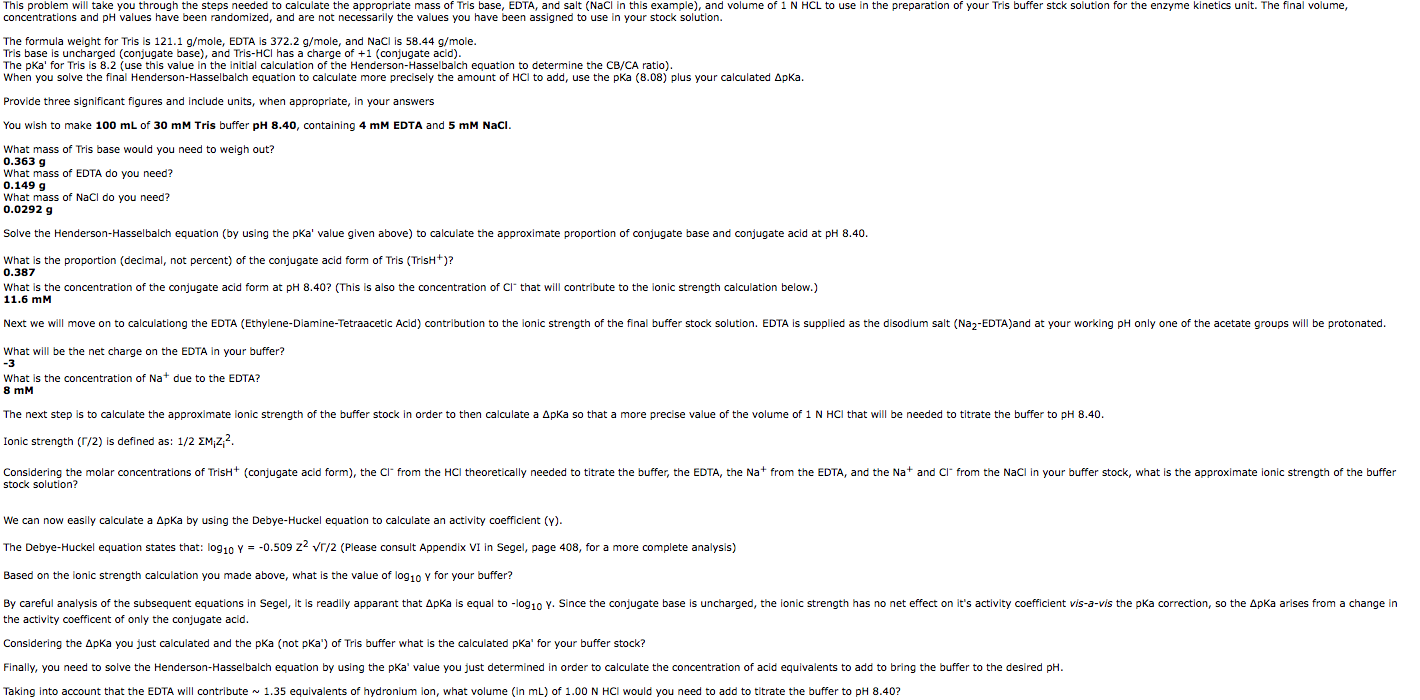
SOLVED: Tris' is a buffer often used in biochemistry. We can simplify the equilibrium reaction as: Tris-HCl + H2O -> Cl- + H3O+ + Tris with pKa = 8.072 and the molar
STOCK SOLUTION RECIPIES: Tris-HCl Buffer 10X Tris-HCl (0.5M Tris Base, pH7.6): Trizma Base ---------------------------------- 6







![Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/11/tris-base-molecular-weight-calculation-300x204.jpg)


![T60040-500.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 500 Grams T60040-500.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 500 Grams](https://d2gdaxkudte5p.cloudfront.net/system/images/T60040-500.0_.jpg)




![Tris Hydrochloride [C4H11NO3.HCl] Molecular Weight Calculation - Laboratory Notes Tris Hydrochloride [C4H11NO3.HCl] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/11/tris-hydrochloride-molecular-weight-calculation-300x225.jpg)